Mechanisms of HSV reactivation
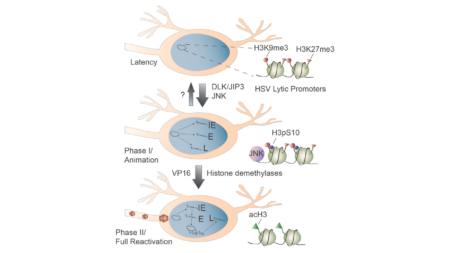
Our lab has developed novel in vitro models of HSV latency. Using these models, we have discovered a neuronal pathway involving activation of c-Jun N-terminal kinase (JNK), common to many stress responses, that is essential for initial HSV gene expression during reactivation. We have also found that this pathway can be activated in neurons by immune-mediated hyperexcitabilty. JNK-dependent reactivation results in a specific change to the HSV epigenome known as a histone methyl/phospho switch, which can permit gene expression to occur from repressive heterochromatin. Our studies therefore answer long-standing question as to how viral gene expression can initiate to trigger HSV reactivation.
Read More